Change of Phase
During a phase change, the additional or removal of thermal energy results in a transition between states (solid, liquid, gas). Phase changes occur at specific temperatures.
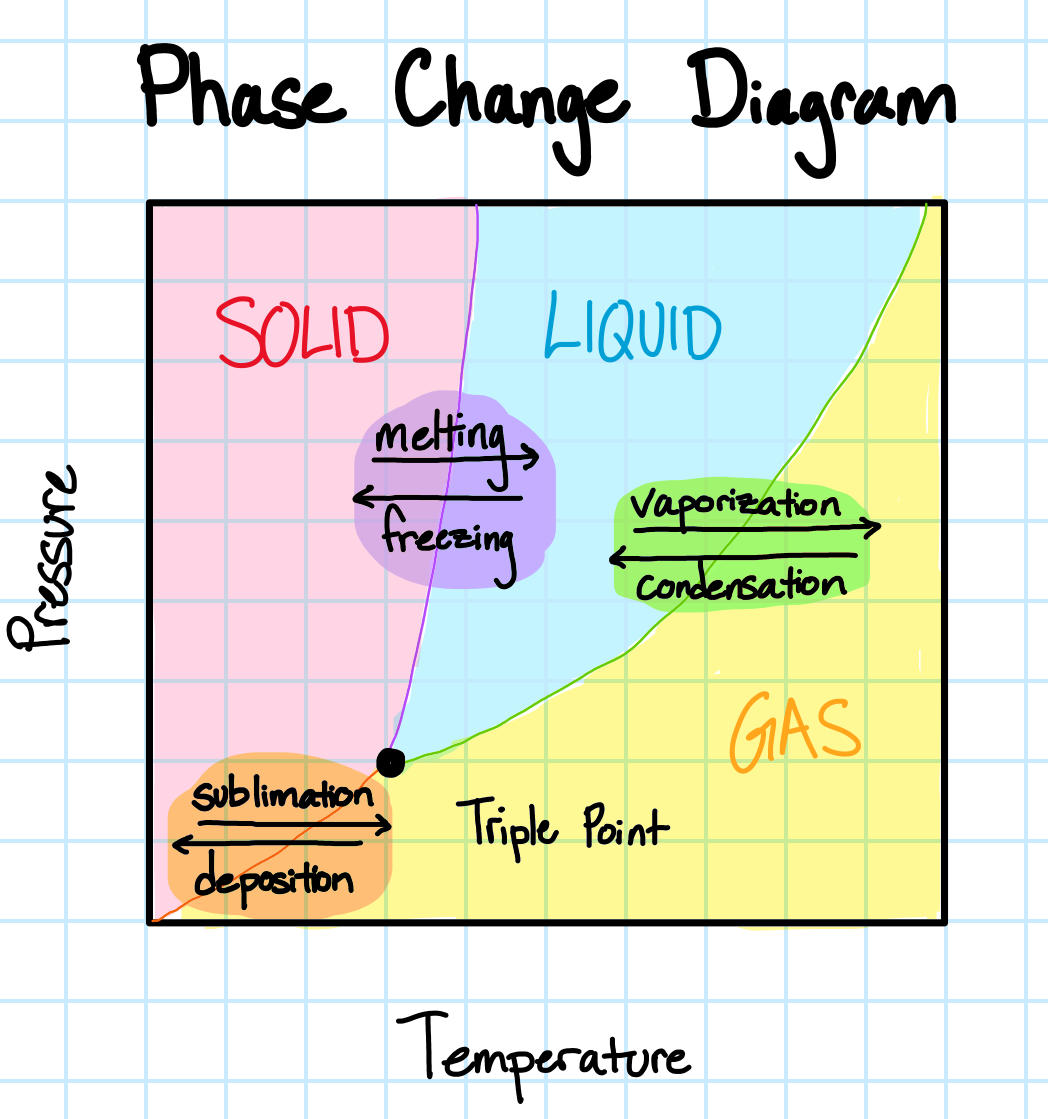
Phase changes complicate heat transfer, as the process is energy intensive and causes changes in the properties of the material. For example, consider the following properties affected with the cooling of water from vapor to liquid to ice:
- Density increases: Solids have the highest density, since atoms are tightly packed together.
- Volume decreases: As the density of a material increases, volume decreases.
- Thermal conductivity increases: The thermal conductivity of ice is nearly three times greater than water.
- Specific heat slightly decreases: Generally, \( C_{p, gas} < C_{p, solid} \leq C_{p, liquid} \). Intuitively, liquids buffer temperature best.
- Dielectric properties (i.e. the interaction of a material with electromagnetic waves) decrease: For example, frozen food takes much longer to microwave, due to poor microwave absorption.
- Elastic modulus decreases: Typically, materials become more brittle at colder temperatures.
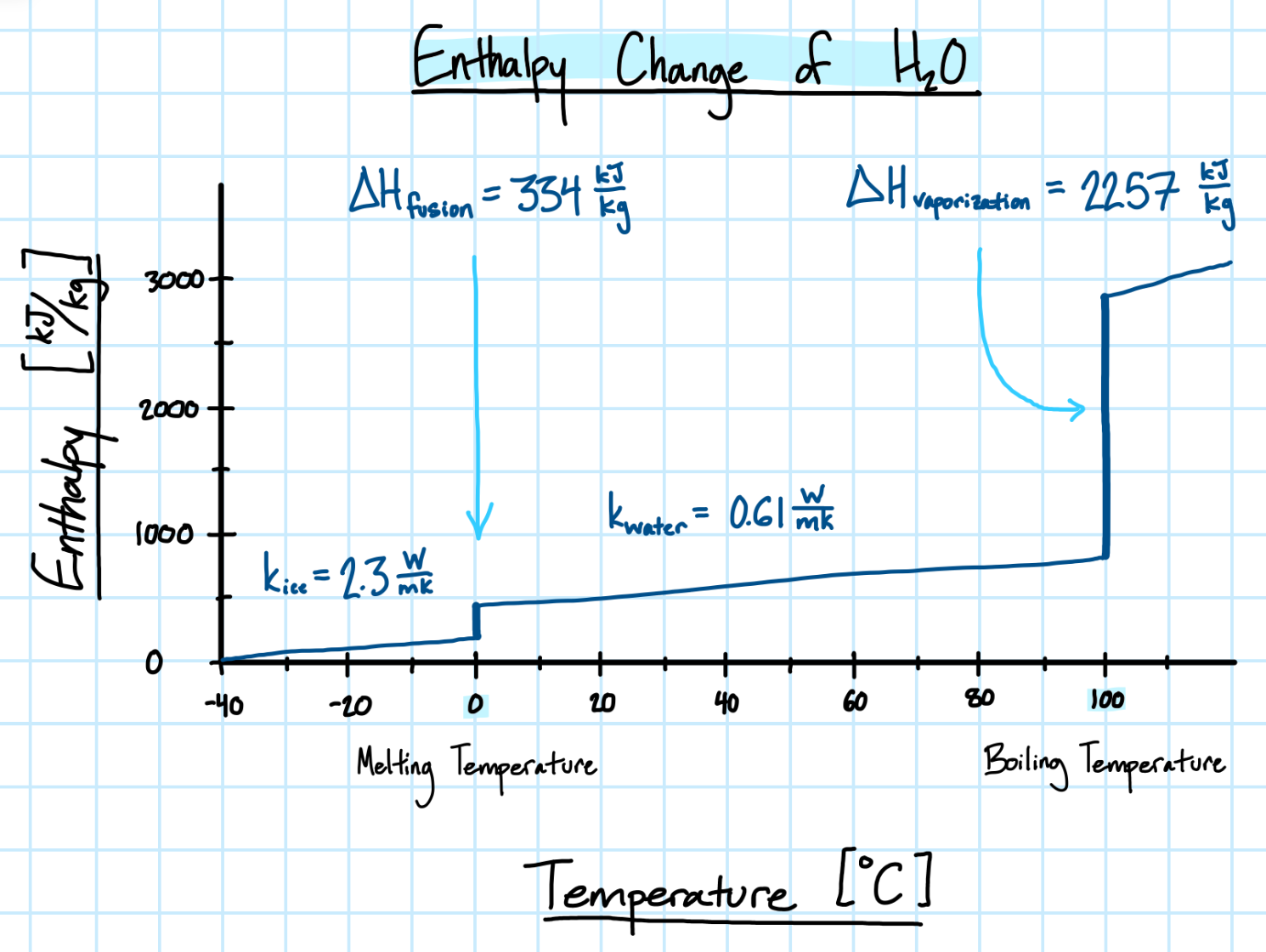
As a material changes phase, so does the energy of the system. The latent heat of fusion (\( \Delta H_{fus} \)) describes the enthalpy difference due to a change of phase from solid to liquid, which occurs at melting temperature. Conversely, the latent heat of vaporization (\( \Delta H_{vap} \)) describes the enthalpy difference due to a change of phase from liquid to vapor, which occurs at the boiling temperature.
Pure Water
Pure water freezes through four sequential stages:
- Supercooling: water cools to less than 0°C before any ice forms. Ultra-pure water may not freeze until −40°C!
- Nucleation: nuclei (crystalline seeds that act as a foundation for ice crystal growth) spontaneously generate.
- Crystal growth: ice crystals grow, controlled by the rate at which heat is removed from the system
- Maturation: smaller ice crystals decrease in size, and larger crystals grow. The number of crystals may decrease, but average crystal size increases!
If the object is frozen from the outside in, the freezing time depends on geometry:
- \( t_f \) = freezing time \( [s] \)
- \( \Delta H_f \) = latent heat of fusion \( [\frac{J}{kg}] \). (Note: for biological tissue with a water component, the latent heat of fusion of water is multiplied by the the water content fraction in the tissue.)
- \( \rho \) = density \( [\frac{kg}{m^3}] \)
- \( T_m \) = melting or freezing temperature \( [K] \)
- \( T_{\infty} \) = surrounding fluid temperature \( [K] \)
- \( L \) or \( R \) = characteristic length, depending on geometry \( [m] \)
- \( k \) = thermal conductivity of the medium \( [\frac{W}{mK}] \)
- \( h \) = convective heat transfer coefficient \( [\frac{W}{m^2 K}] \)
Looking at key parameter relationships, freezing time decreases with
- higher thermal conductivity \( k \) (less resistance to heat flow)
- higher convective coefficient \( h \) (stronger surface cooling)
- larger temperature difference \( \Delta T \) (stronger driving force)
- smaller characteristic length \( L \) (there is more mass to freeze)
The frozen layer thickness grows as \( x \propto \sqrt{t} \); freezing progressively slows as the frozen layer thickens because it adds thermal resistance.
Need a review?
Be sure to use the appropriate characteristic length for the geometry!
Heterogeneous Solutions
Solutions behave differently than pure water. Because solutes are present, heterogeneous nuclei form more readily, so nucleation occurs earlier and less supercooling is required.
When solutes (e.g., salt) dissolve in a solvent (e.g, water), they interfere with hydrogen bonding, depressing the freezing point below 0°C. The relationship is expressed as:
- \( \Delta H_{f} \) = latent heat of fusion of pure liquid \( [\frac{J}{kg}] \)
- \( R_{g} \) = ideal gas constant, \( 8.314 [\frac{J}{mol K}] \)
- \( T_{pure} \) = freezing point of pure liquid \( [K] \)
- \( T_{soln} \) = freezing point of the solution \( [K] \)
- \( x_{A} \) = mole fraction of solvent A (e.g., water) in solution
As freezing proceeds, water crystallizes out of solution, leaving behind a progressively more concentrated liquid, which in turn depresses the freezing point further. This process continues until all freezable water has solidified. Practically, this means biological solutions freeze gradually over a temperature range (roughly −2 to −35°C) rather than at a single sharp temperature.