Zeroth Law: Thermal Equilibrium
The zeroth law of thermodynamics establishes the concept of thermal equilibrium and provides a foundation for defining and measuring temperature. It states that if two systems are in thermal equilibrium with a third, then they are in thermal equilibrium with each other. If a temperature difference exists, then heat will flow (from hot to cold) only until thermal equilibrium is reached. Therefore, the zeroth law essentially defines the condition under which we expect heat transfer to cease.
This law plays a crucial role in understanding heat transfer by identifying that temperature is a shared property of systems in equilibrium and explaining why it can be compared consistently between different objects. It also provides the physical justification for the use of thermometers. When they are placed in contact with an object and allowed to reach thermal equilibrium, the thermometer and the object share the same temperature.
Remember!
Temperature and heat are related, but not the same thing! Temperature is a property of an object or system that describes the average kinetic energy of the particles within it. Heat refers specifically to the transfer of thermal energy across a system boundary due to a temperature difference.
First Law: Conservation of Energy
The first law of thermodynamics expresses the principle of energy conservation: energy cannot be created or destroyed, and can only be transferred or converted between forms. This law helps in understanding all situations of heat transfer and explains why temperature change occurs.
A system’s temperature can increase only if thermal energy enters the system or is generated internally, and it can only decrease if thermal energy leaves the system or is consumed. Temperature changes as a consequence of energy transfer or conversion, and never spontaneously.
When the first law is applied to heat transfer problems, it is expressed as a balance within the boundary of the system: energy entering the system, energy leaving the system, generation or consumption of heat within the system, and energy stored within the system.
Second Law: Directionality of Heat Transfer
The second law of thermodynamics is important in understanding the directionality of heat transfer. It states that in an isolated system, the total energy of the system can only increase or remain constant over time.
In heat transfer, this explains why heat transfer only spontaneously occurs from regions of higher temperature to regions of lower temperature. This type of transfer increases the total entropy of the system in alignment with the second law.
Conduction
Conductive (or diffusive) heat transfer is the transport of thermal energy through a medium due to temperature gradients within the material. Energy is transferred down the temperature gradient from high to low. The net transfer of energy is due to random molecular motion.
where
- \( q^{''}_x [\frac{W}{m^2}] \) = flux, or rate of heat flow per unit area
- \( q_x [W] \) = rate of heat flow in the x direction
- \( A [m^2] \) = surface area perpendicular to the direction of heat flow
- \( T [K] \) = temperature of the medium at x
- \( k [\frac{W}{mK}] \) = thermal conductivity of the medium
Remember!
Always keep track of your units. To convert between Fahrenheit, Celsius, and Kelvin:
Steady State
In steady state heat conduction, temperature does not change with time. However, temperature can change with position, and this gradient drives the transfer of heat. There are three main examples of steady state heat conduction:
- In a slab
- In a cylinder
- With heat generation (Q)
Unsteady State
In unsteady state (or transient) heat conduction, temperature changes over time. There are three main examples of unsteady state heat conduction:
- With no spatial variation
- With semi-infinite geometry
- With finite geometry
Convection
where
- \( q_{1-2} [W] \) = rate of heat flow from 1 (hotter) to 2 (colder)
- \( A [m^2] \) = surface area normal to the direction of heat flow
- \( T_1 [K] \) = temperature of 1 (e.g., system)
- \( T_2 [K] \) = temperature of 2 (e.g., surroundings)
- \( h [\frac{W}{m^2 K}] \) = convective heat transfer coefficient (which always includes the effect of conduction in the fluid)
Note that the convective heat transfer coefficient is not a material property.
Radiative Heat Transfer
Radiative heat transfer is the transport of thermal energy emitted as electromagnetic waves. It is attributed to changes in electron configuration within the atoms of the target matter. No medium (i.e. physical contact or fluid motion) is required for this mode of heat transfer, as it is spontaneously emitted.
where
- \( q^{''} [\frac{W}{m^2}] \) = rate of heat flow per unit area
- \( q [W] \) = rate of heat emission
- \( A [m^2] \) = area of radiating surface
- \( \sigma = 5.67*10^{-8} [\frac{W}{m^2 K^4}] \) = Stefan-Boltzmann constant
- \( T [K] \) = temperature of the radiating surface
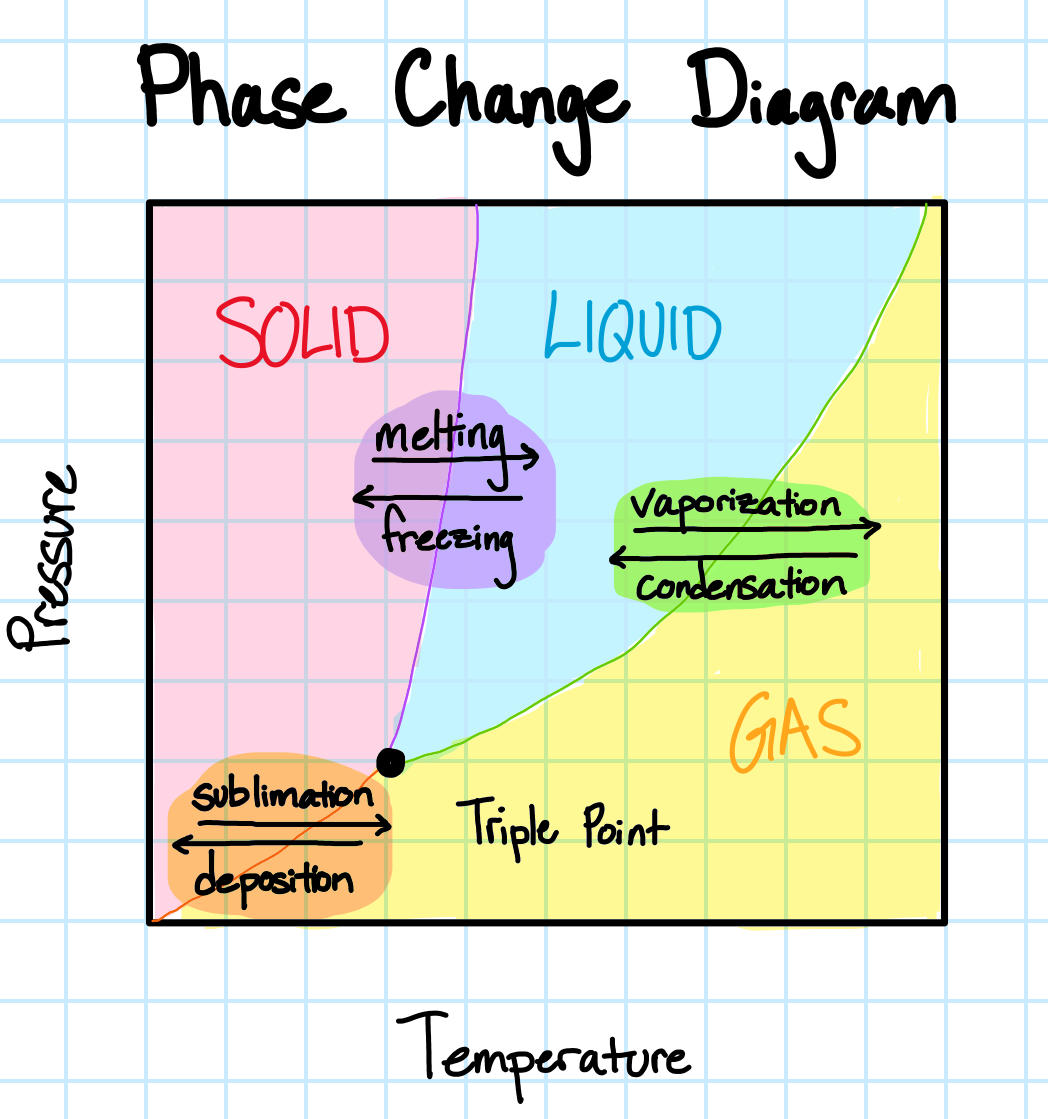
Heat Transfer with Phase Change
Phase changes complicate heat transfer, as the process is energy intensive and causes changes in the properties of the material. Affected properties include volume, density, thermal conductivity, specific heat, dielectric properties (i.e. the interaction of a material with electromagnetic waves), and mechanical properties (i.e. elastic modulus)
For example, consider the cooling of water from vapor to liquid to ice:
- Density increases. Solids have the highest density, since atoms are tightly packed together.
- Volume decreases. As the density of a material increases, volume decreases.
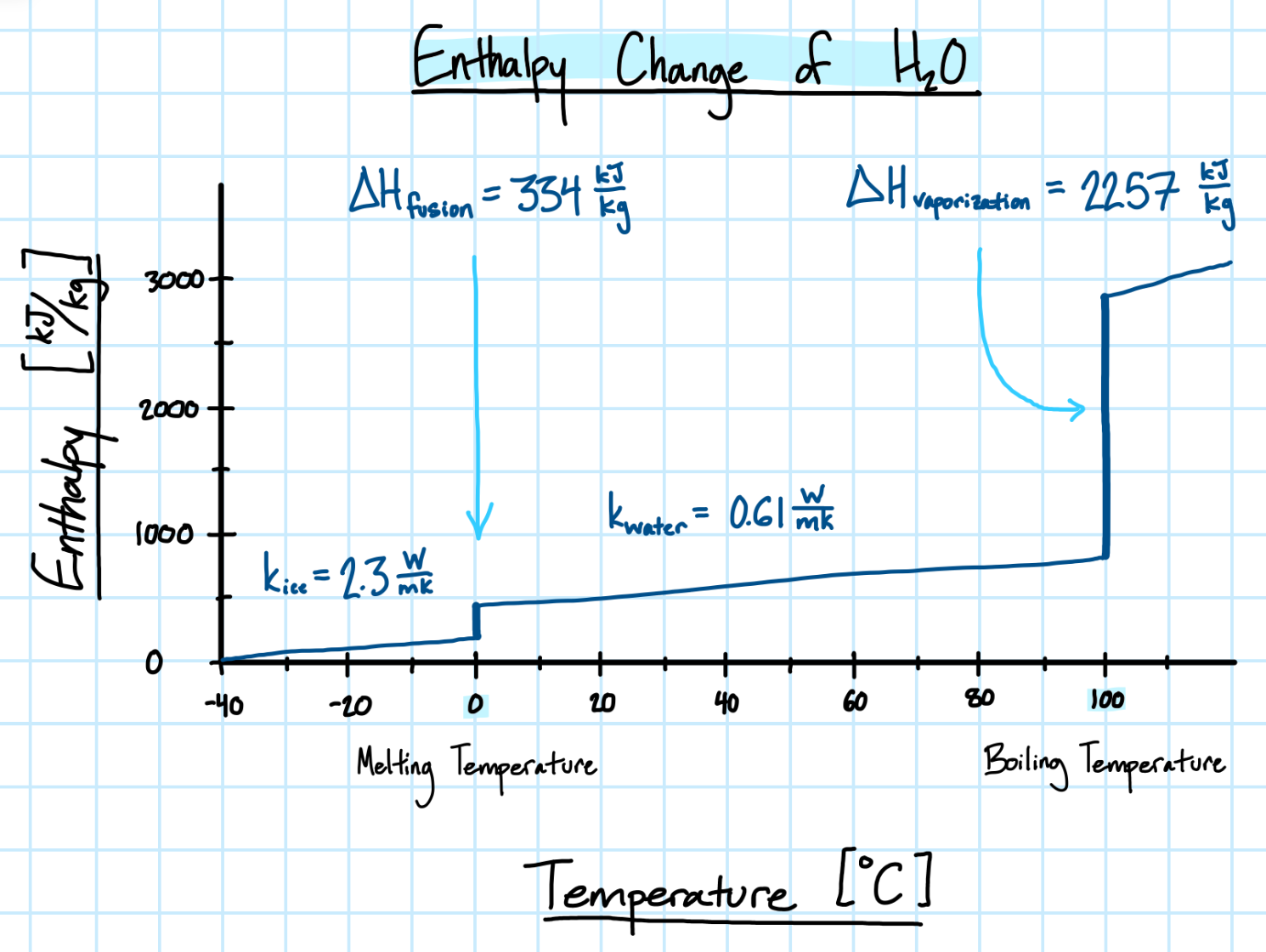
- Thermal conductivity increases. The thermal conductivity of ice is nearly three times greater than water.
- Specific heat slightly decreases. Generally, \( C_{p, gas} < C_{p, solid} \leq C_{p, liquid} \) Intuitively, liquids buffer temperature best.
- Dielectric properties decrease. For example, frozen food takes much longer to microwave, due to poor microwave absorption.
- Elastic modulus decreases. Typically, materials become more brittle at colder temperatures.
As a material changes phase, so does the energy of the system. The latent heat of fusion (\( \Delta H_{fus} \) ) describes the enthalpy difference due to a change of phase from solid to liquid, which occurs at melting temperature. Conversely, the latent heat of vaporization (\( \Delta H_{vap} \) ) describes the enthalpy difference due to a change of phase from liquid to vapor, which occurs at boiling temperature.
General Heat Transfer Governing Equation
The governing heat equation is derived from an energy balance applied to a differential control volume and Fourier's Law. It includes all possible mechanisms that can contribute to heat transfer in a system, and encapsulates the combined effects of energy storage, conduction, convection, and internal heat generation, and allows the prediction of the temperature profile within the system.
The governing heat equation can be written in Cartesian or radial coordinates. While heat transfer can occur in three dimensions, it is simplified here to be solved with heat flowing only in one direction (x,r).
In practice, many problems involve simplifying assumptions (e.g., steady state, no fluid flow, or no internal heat generation), which allow certain terms to be removed. The meaning of each term and the conditions under which it should be retained or dropped are summarized in Table 1.
Table 1 here...
Boundary and Initial Conditions
To obtain a particular solution, we must solve the general governing equation utilizing known quantities called boundary conditions (which describe heat transfer at particular surfaces and interfaces) as well as initial conditions (for unsteady state or time-dependent problems).